Hello everyone, welcome to our monthly newsletter, April 2026 edition.
Exciting Announcement
BG published probably the most important paper of his career : “How I Read a Clinical Trial Report? A Primer for Busy Clinicians” in JCO Oncology Practice. This paper has went “viral” with 275K views on X/Twitter and over 17,000 downloads on the journal page within a week of publication. Most readers have commented that this fills one of the most important gaps in medical education- we tell trainees that they need to read trial reports, but don’t teach them how to. BG states that “the real utility of this paper would be if many people do start reading clinical trials more critically using this primer and if it stood the test of time such that trainees from many years later would continue to use this for reading clinical trials.”
Although written as “a primer for busy clinicians,” several patient advocates have highlighted that this is also a useful resource for patients and advocates to understand clinical trial reports. Thankfully, this paper is published as open-access, and thus should be readily accessible to patient community.
Read our news post about this paper, and the full article!
Publications
- The above-discussed “How I read a paper?” was our most important publication last month, but we have more!
- We are very excited to share this comment published in Nature Reviews Clinical Oncology: “Did ATOMIC really improve on the standard of care for mismatch repair-deficient colon cancer?”. We are excited about this paper because:
- This is the first Nature Reviews paper for our fellow Javier-David Benitez-Fuentes. Icing on the cake, he is the first-author. As you know from our March Newsletter, he joined our lab only last month, and now he already has his first Nature Reviews paper as the first-author. At BG Lab, we really are excited about our trainee’s milestones like this, and celebrate such milestones.
- We hopefully inform policy and practice regarding the implications of the ATOMIC trial, specifically regarding overtreatment, treatment de-escalation, surrogacy, post-trial access to treatment and contributions of component. In this paper, we argue that the ATOMIC trial should inform future clinical trials testing de-escalation rather than changing current clinical practice. For people who have read our “How I read a paper” paper, this ATOMIC comment provides an example of applying those critical appraisal skills in real-life.
- BG gets to combine his passion of Zen spirituality and academic discourse in this article. He uses a Zen Koan as an analogy of the evolution of adjuvant treatment of stage 3 colon cancers. Please read, we assure you you will like it. If you don’t have access, email us and we will send you a PDF. Or, just follow us on X!
- BG, along with Research Assistant Brian Shkabari and other colleagues from Common Sense Oncology Patient Priorities Working Group, published a new article in JNCI: “Common sense oncology values to action: a patient led framework for randomized clinical trial design.” There is rising concern that randomized clinical trials (RCTs) do not adequately prioritize outcomes that matter to patients. This framework is a practical tool that can promote more ethical and relevant clinical trials for patients on a global scale.
Trainee and Mentee News

- BG’s Research Assistant, Claire Diana-Gonsalves, gave her first-ever(!) oral presentation on the Landscape of Solid Cancer Drug Reimbursement Decisions by Canada’s Drug Agency at the Cancer Care and Epidemiology Seminar on April 23rd.
Media

- BG Lab released their third Grounded in Groundshot podcast episode with special guest Professor Chris Jackson. This episode is a must-watch! BG and Dr. Jackson discuss what the definition of success is, specialization versus general oncology in high-income versus low-income countries, and health technology assessment systems and how patient advocacy groups can influence health policy and frameworks. Dr. Jackson is not only BG’s close friend, but a strong influence on cancer policy in New Zealand. Watch this episode on Youtube, or listen on Spotify or Apple Podcasts.
- BG also chatted with Dr. Dario Trapani for the TJ Talks podcast about cancer policy, mentorship, outcomes that matter, cancer Groundshot, BG Lab, and our own Grounded in Groundshot podcast! Listen here, it is a fun conversation. https://omny.fm/shows/tumori-journal/outcomes-that-matter-a-common-sense-revolution-in-oncology
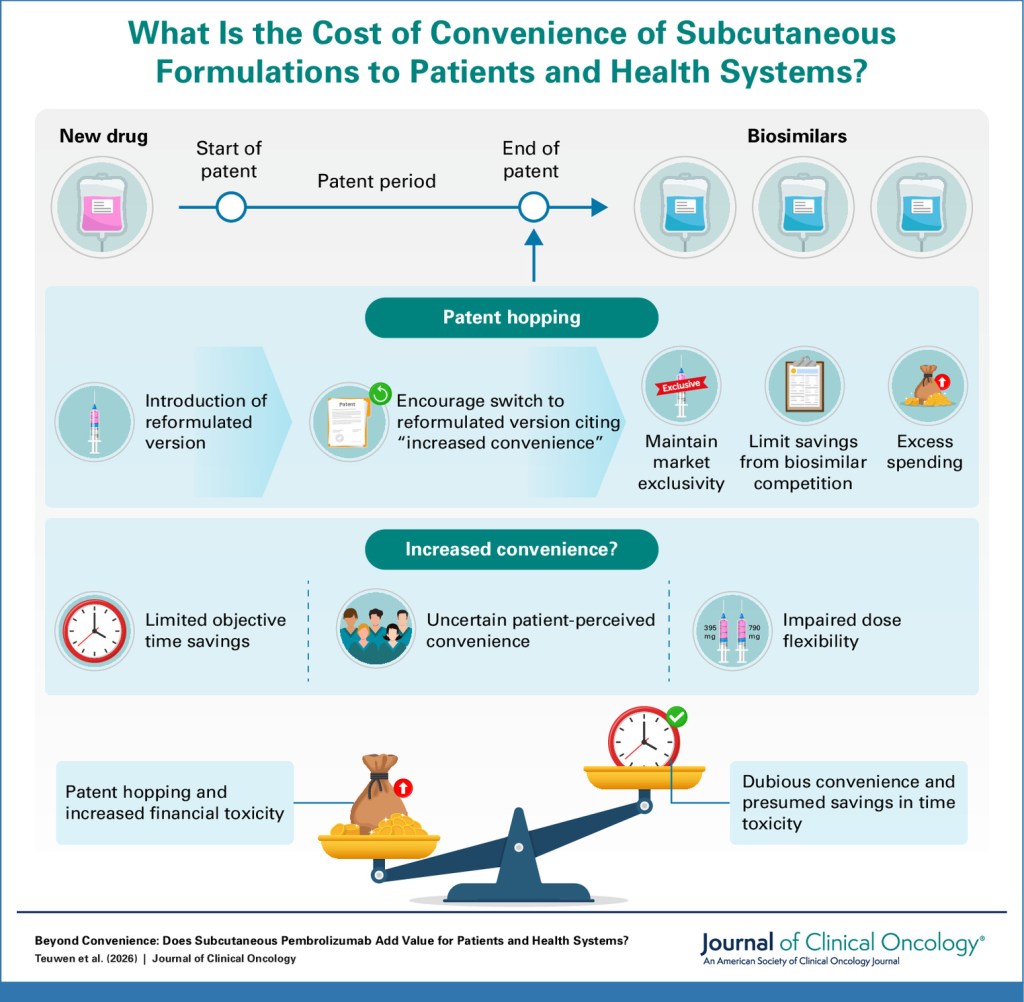
- Last month, we highlighted a new publication about the cost of subcutaneous pembrolizumab for patients and health systems. This month, BG had a conversation with GU Oncology Now about pembrolizumab’s clinical benefit and alignment with patient values, and spoke about the broader global and regulatory context in relation to “patent hopping.” Oncology News Central also ran a news article about this, titled “Subcutaneous Pembrolizumab: Meaningful Advance or Pseudo-innovation” based on an interview with BG. Read the original publication here, and read the interview for a better understanding of this debate.

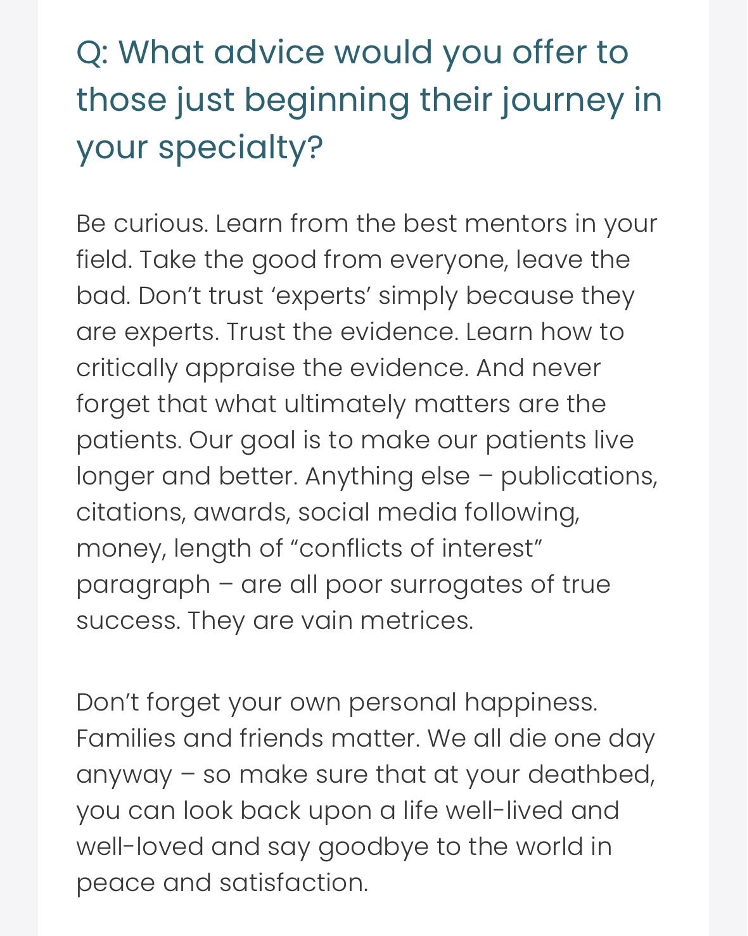
- BG was announced as a touchONCOLOGY Future Leader 2026 – someone who is a “rising star” that will shape the future of policy and patient care for gastrointestinal oncology. Read about what work BG feels is most fulfilling, how he became interested in his speciality, and advice he would give to those beginning their journey in medicine in his Future Leaders interview. A snippet of his advice is below, which has resonated with many others.
- BG was quoted in an article by the International Consortium of Investigative Journalists regarding pricing concerns with Keytruda, Merck’s blockbuster drug. Read the full article here.
- BG released a new Medscape Skills Lab video, Beyond P Values: Defining True Clinical Effect Size. In this video, he discusses the nuances of sample size, statistical significance, clinical meaningfulness, and absolute versus relative risk reduction. Watch it here!
- BG released another Medscape Skills Lab video, dissecting the FDA’s decision to deny approval for Replimune’s cancer drug RP-1 for advanced melanoma. Watch it here. He goes deeper about the value of response rates as the basis for drug approval, and nuances of how those responses can be misleading based on timing of intratumoral injections in relation to time of assessment.
- BG was listed as one of the most active oncologists for lung cancer discussion during European Lung Cancer Congress.
Talks, Presentations, and Meetings

- BG Lab is super proud that our patient partner Dr. Sharon Batt presented a poster at AACR Annual Meeting sharing our results regarding trends in number, evidence standards, and magnitude of benefit of cancer drugs approved by Health Canada. We hope that this sets an example that it’s not always the PIs or even the students who should go to conferences to present the results- sometimes it can be the patient partners!


- BG was busy hosting/participating/talking at the CSO Annual Leadership meeting in Montreal.

- BG was in Montreal again a week later to participate in the CIHR Institute of Cancer Research IAB meeting where he contributed to rich discussions on strategic directions and priorities for Canadian cancer research.
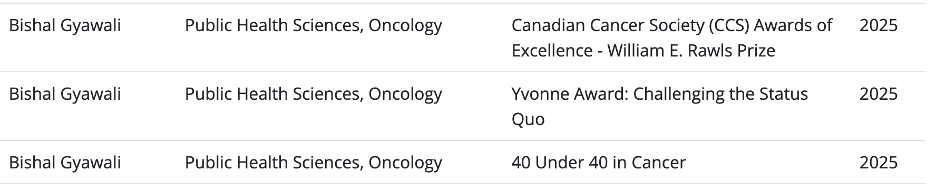
On April 9, BG was felicitated by Queen’s University among other Queen’s Faculty who have received a major national or international award in the last 2 years. The program titled “Applause: A Celebration of Research Excellence” is organized by Queens University VP Research office every 2 years. BG was delighted to represent Faculty of Health Sciences by receiving these 3 major awards last year. His family joined him for the event.

Upcoming Events
- Claire Diana-Gonsalves will be giving an oral presentation on the Landscape and Trends of Solid Cancer Drug Reimbursement Decisions by Canada’s Drug Agency at the CAHSPR conference in May! This will be her first conference. BG will be in the audience supporting Claire!
- BG will be attending the 2026 ASCO Annual Meeting from May 29th to June 2nd in Chicago. ASCO Annual Meeting is the busiest season for BG and BG Lab. Meet BG in the following sessions:
- He is the Discussant of Oral Abstracts for the Care Delivery/Models of Care/Global Oncology track. Find him in this session on Monday, June 1, 3-6 pm at Room S100bc.
- He will be presenting the awards at the Health Policy Community of Practice session on May 30 Saturday, 8-9:30am in Room E253cd.
- Same day, 9am to 12 noon, he will be presenting a poster of our work on access to adjuvant chemotherapy and outcomes of colorectal cancer for immigrant versus non-immigrant patients in Ontario.
- May 29 Friday 4-6pm he will be at the Grants and Awards ceremony, because one of our fellows….let’s keep it a secret! We will inform you when the embargo on this good news is lifted!
- Friday night, he – and our other members of BG Lab, Drs. Teuwen and Mina- will be at the Oncodaily party because there is something pretty big and pretty cool happening. We will give you a hint- BG lab and Oncodaily are collaborating! How, what, why? Go there to find out!
From the Archives
One of the first research methods that BG learned (self-taught 😊) was meta-research, including systematic reviews and meta-analysis. He started employing these methods to study the harms of cancer drugs. He first studied the risks of fatal and serious adverse events with sorafenib which was published in Annals of Oncology. When adjuvant sunitinib had conflicting results in kidney cancer, he conducted meta-analyses of these conflicting trials to conclude that adjuvant sunitinib should not be considered practice changing, also published in Annals of Oncology. He used these data further to make an argument against FDA’s decision to approve adjuvant sunitinib, and published the famous regulatory capture paper in JAMA Oncology. These papers were cited by several guidelines that did not endorse adjuvant sunitinib despite FDA approval. This was his first experience of influencing policy through academic publications.
He continued to use these methods to study risk-benefit profiles of other cancer drugs such as ramucirumab, and relative efficacy versus harms of different oral anticoagulants to prevent and treat venous thromboembolism in patients with cancer.
However, BG does not do much meta-analysis these days because he thinks these methods are now being misused and abused. He advocates for thoughtful meta-analyses to answer important questions in policy or practice, not meta-analysis just for the sake of publishing an article. There are a lot of garbage meta-analyses in the literature. BG published this viewpoint in The Lancet Oncology later titled “Meta-analyses and RCTs in oncology—what is the right balance?” which is now a classic resource used to teach why it does not make sense to do meta-analysis about everything.
Do you like what we do? You can support us!
The type of work that we do is often not funded by traditional funding channels. If you like our work and want to support us with a donation to our lab, please reach out directly to BG at gyawali.bishal@queensu.ca.
If you want to make a one-time contribution or regular contributions, please click here.
Please subscribe to our newsletter, X account, Youtube channel, and spread the word.
Thank you for reading and stay tuned for next month’s update!

Leave a comment